FAQs about PNH causes
Paroxysmal nocturnal hemoglobinuria (PNH) and aplastic anemia are both forms of bone marrow failure disorders, which affect stem cells in the bone marrow that are responsible for giving rise to new blood cells. PNH and aplastic anemia often co-occur: it is estimated that up to 25% of aplastic anemia patients will develop PNH, and up to 40% of those with PNH will develop aplastic anemia at some point. Each disorder is a risk factor for the other.
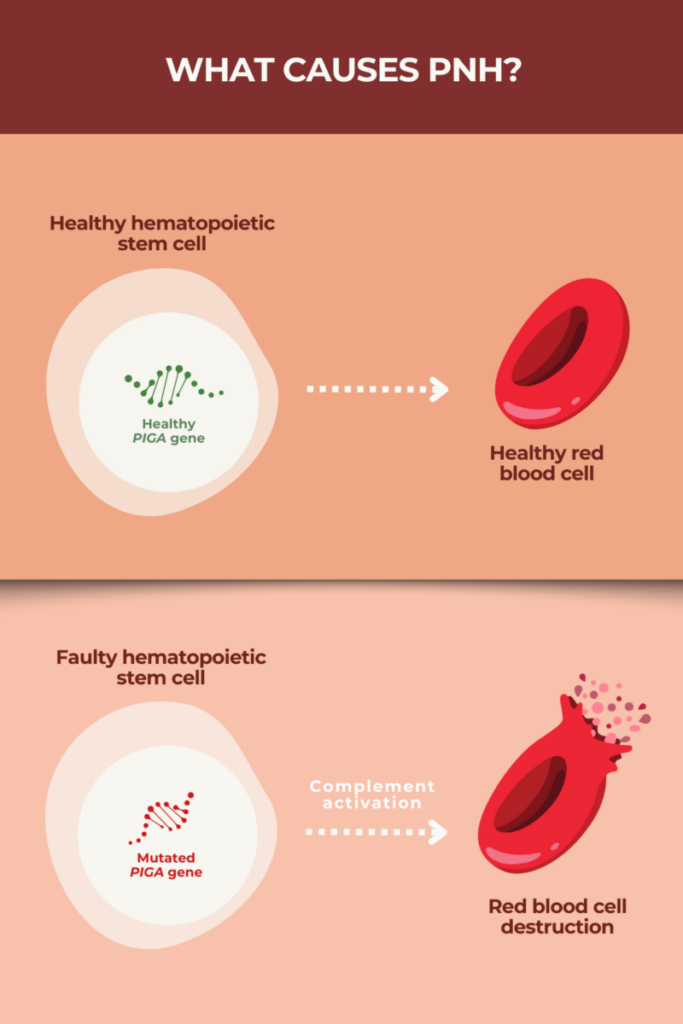
Most cases of paroxysmal nocturnal hemoglobinuria (PNH) are caused by mutations in the PIGA gene in specific stem cells responsible for producing new blood cells in the body. A person is not born with the mutation that causes PNH; instead, the mutation develops randomly due to an error in stem cell division during a person’s lifetime.
Paroxysmal nocturnal hemoglobinuria (PNH) can cause life-threatening complications, particularly blood clots that can disrupt blood flow (thrombosis). However, with modern therapies, life expectancy for someone with PNH is generally comparable to that of the general population.
The risk of paroxysmal nocturnal hemoglobinuria (PNH) is increased in people with another bone marrow condition called aplastic anemia. Around 15% to 25% of aplastic anemia patients will go on to develop PNH. Other risk factors for PNH have not been identified.
Thrombosis, a condition in which blood clots form and block blood flow, is the most common fatal complication of paroxysmal nocturnal hemoglobinuria (PNH). In PNH, the dysfunction of platelets (small cell fragments in the blood that stop/prevent bleeding) and low levels of nitric oxide can set the stage for abnormal blood clotting, which may ultimately lead to thrombosis.
 Fact-checked by
Fact-checked by